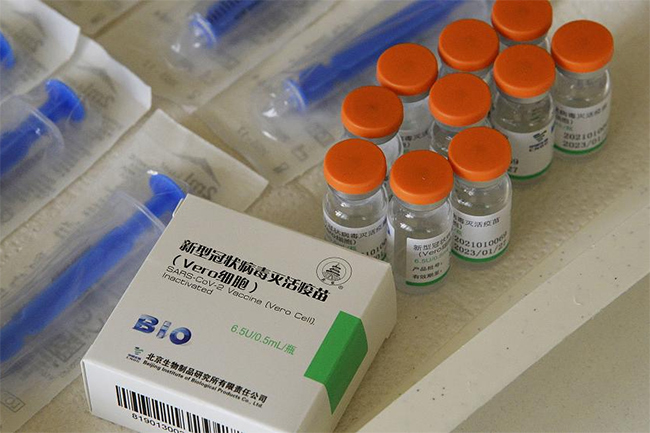
The expert panel of Sri Lanka’s National Medicines Regulatory Authority (NMRA) has approved the emergency use of China’s Sinopharm COVID-19 vaccine in the country, says State Minister Prof. Channa Jayasumana.
Meanwhile, Sri Lanka began rolling out the Sinopharm jabs among the locals this afternoon (May 08).
The Sinopharm jabs are administered based on the coronavirus cases identified in each Grama Niladhari division.
A consignment of 600,000 doses of the vaccine donated by China arrived on the island on March 31.
Chinese nationals residing in Sri Lanka were given priority under the first phase of the Sinopharm vaccination drive in the country.
Sinopharm vaccine, also known as BBIBP-CorV, is produced by China National Biotec Group (CNBG) – a subsidiary of China National Pharmaceutical Group Corporation (SINOPHARM).
The World Health Organization (WHO) on Friday (May 07) gave the nod for emergency use of China-made Sinopharm jabs across the world.
It became the first COVID-19 shot developed by a non-Western country to win the WHO’s approval.



































